

Five-year data now available

Take a closer look at HEMGENIX efficacy, safety, and
durability at 5 years.1
Take a closer look at HEMGENIX efficacy, safety, and durability at 5 years.1
Connect with us
Meet with your CSL Behring Associate Director of Corporate Accounts.
Schedule a meetingFormulary kit
Review product-specific resources for the first gene therapy for hemophilia B.
About hemophilia B
Hemophilia B is a rare, X-linked hereditary disorder characterized by bleeding, pain, and long-term complications.2-4 Hemophilia B is caused by a defect in a gene encoding coagulation factor IX, which is expressed primarily in hepatocytes. This mutation occurs in 1 in every 20,000 to 25,000 male births.4-6 The severity of hemophilia B typically correlates to native coagulation factor activity. Severe hemophilia B is characterized by spontaneous and/or traumatic bleeding into joints, muscles, and internal organs and can result in reduced life expectancy.3,4
What is HEMGENIX?
HEMGENIX is an adeno-associated virus vector-based gene therapy indicated for the treatment of adults with hemophilia B (congenital factor IX deficiency) who:
- Currently use Factor IX prophylaxis therapy, or
- Have current or historical life-threatening hemorrhage, or
- Have repeated, serious spontaneous bleeding episodes.
HEMGENIX is for single use intravenous infusion only.
What are the key benefits?
At 5 years, a single dose of HEMGENIX is associated with1:
REDUCTION IN MEAN
FACTOR IX CONSUMPTION
FROM LEAD-IN PERIOD

MEAN FACTOR
IX ACTIVITY*

REDUCTION IN ANNUALIZED
BLEED RATE (ABR)
vs routine factor IX
prophylaxis†
*Elevated factor IX levels have been observed annually (n=48).
†Primary endpoint demonstrating noninferiority of ABR during months 7 to 18 compared with lead-in period was met. Noninferiority comparison and mean ABR estimates were based on a repeated measures generalized estimating equations negative binomial regression model (P=0.0035).
The prospective, single-dose, single-arm study included a total of 54 patients aged 19 to 75 years. Patients completed a ≥6-month observational lead-in period. Adjusted ABR for all bleeds decreased from an average of 4.16 for prophylaxis during the lead-in period to 1.52 in months 7 to 60 after treatment, a reduction of 63%. During months 7 to 18, ABR for all bleeds decreased by 64%.
HEMGENIX offers elevated and sustained factor IX levels for years after a single infusion1
At 5 years, HEMGENIX continues to:

Be associated with NO serious treatment-related adverse reactions
NOT be associated with the development of inhibitors to factor IX
Mean factor IX activity sustained at 36% at 5 years after single HEMGENIX infusion1
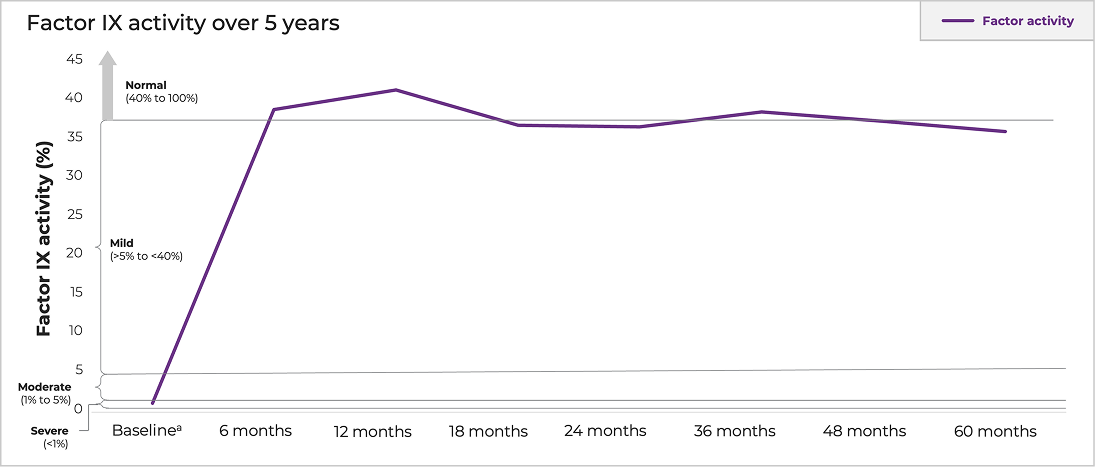
No clinically meaningful correlation was identified between a subject’s AAV5 NAb titer at baseline (up to a titer of 1:678) and their factor IX activity at year 5 post dose1
Abbreviation: ICER, Institute for Clinical and Economic Review.
*Baseline factor IX was imputed based on the subject’s historical hemophilia B severity documented on the Case Report Form. If the subject had documented severe factor IX deficiency (factor IX plasma level < 1%), their baseline factor IX activity level was imputed as 1%. If the subject had documented moderately severe factor IX deficiency (factor IX plasma level ≥1% and ≤2%), their baseline factor IX activity level was imputed as 2%. Standard error was not provided at baseline.7
Uncontaminated data from the central laboratory were used; “uncontaminated” meant that the blood sampling did not occur within 5 half-lives of exogenous factor IX use. Both the date and time of exogenous factor IX use and blood sampling were considered in determining contamination. Factor IX levels beginning with the week 3 assessment were used in the analysis. All efficacy data collected after liver transplants were excluded from the analysis.7
Demonstrated reduction of factor IX utilization at 5 years1

from lead-in period to 5 years
post treatment
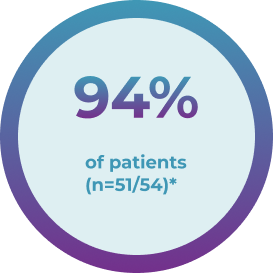
eliminated routine factor IX prophylaxis and remained prophylaxis free through 5 years post treatment
*Two patients experienced lack of efficacy. One patient had the highest NAb titer of 1:3212, and 1 patient received ~10% of the planned dose. One patient returned to factor IX prophylaxis at month 29 and their last factor IX activity levels were 3.6%. An additional patient required intermittent prophylaxis for approximately 20 weeks during months 7 to 18.
At 5 years, the HEMGENIX safety profile was consistent, with no new treatment-related AEs1
Common treatment-related adverse events (TRAEs) reported in >8% of patients (safety population)
| TRAEs by MedDRA preferred terma | Up to Year 5 | |
|---|---|---|
| n (%) | Number of Events | |
| At least 1 TRAEb | 39 (72.2) | 100 |
| ALT increased | 10 (18.5) | 11 |
| Headache | 8 (14.8) | 9 |
| Influenza-like illness | 7 (13.0) | 8 |
| AST increased | 6 (11.1) | 7 |
Three patients had infusion-related reactions that required a dose interruptionc
Abbreviations: AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase; MedDRA, Medical Dictionary for Regulatory Activities.
aMedDRA Version 26.0 was used for coding.
bIncludes possibly related or related.
cInfusion-related reactions were defined as any AEs related to the investigation of medical product administration procedures or unexpected reactions. Infusion reactions were any treatment-emergent AE occurring within 24 hours of infusion, qualifying for special notification, assessed as related or possibly related by the investigator, and considered as an infusion-related reaction during the safety assessment. They were infusion-site reaction, hypersensitivity (ie, urticaria), facial flushing, itching, headache, dizziness, etc.
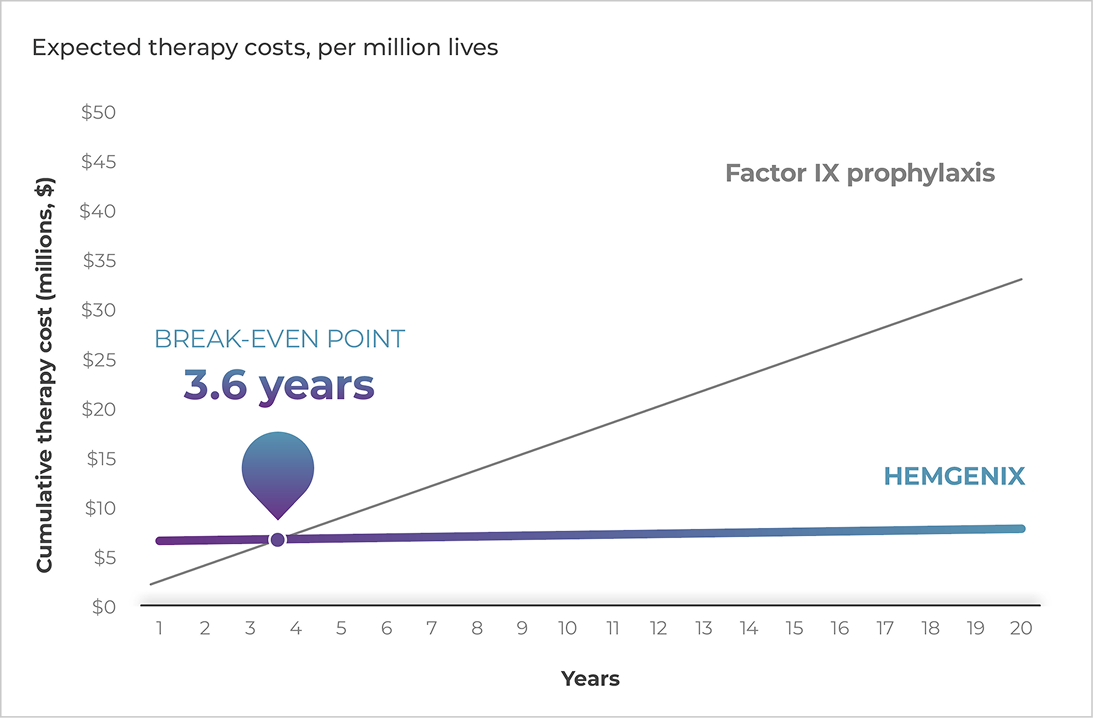
HEMGENIX is estimated to start generating cost savings at 3.6 years8*
Modeled savings with HEMGENIX: gene therapy vs factor IX prophylaxis†
Payer retention among hemophilia B patients is strong—in a retroactive analysis of 666 patients, the average member stayed with their plan for 5.2 years9
*Based on the HEMGENIX Access Decision Model. Accessed November 2025.
†Published wholesale acquisition cost (WAC) price.
HEMGENIX offers greater potential savings compared with subcutaneous options10*
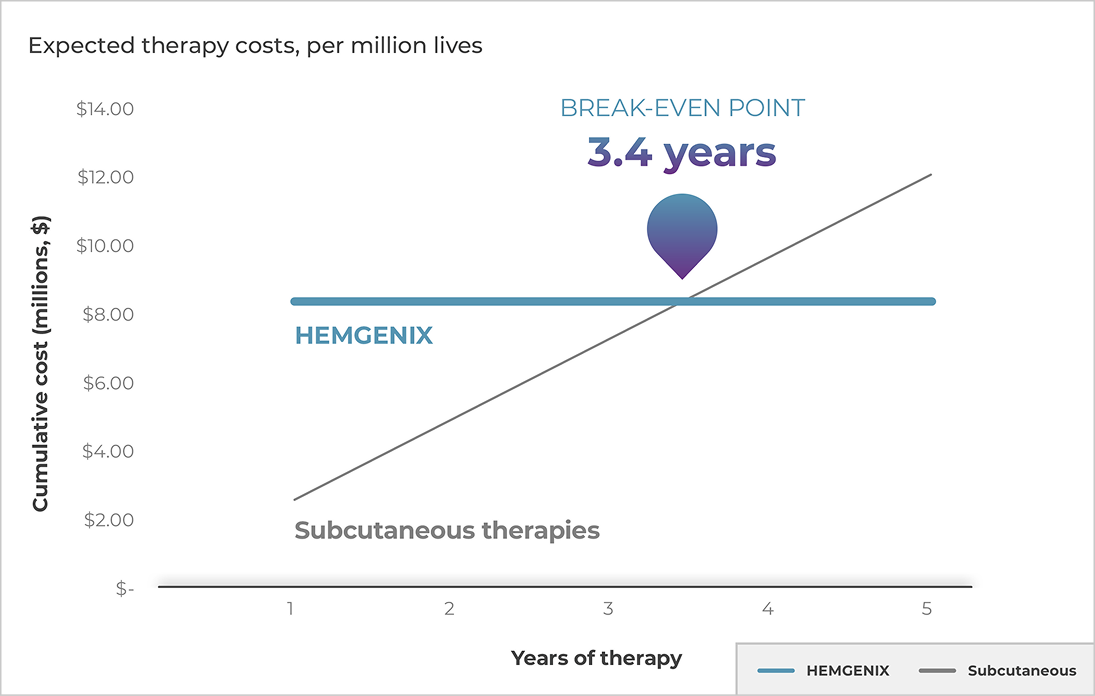
Using cost of drug therapy alone, plans could save $3.85 million over the course of 5 years if their indicated patients use HEMGENIX instead of subcutaneous therapies
*Based on WAC, treatment regimen, and a 93.9 kg patient where appropriate.
Want to learn more?
Meet with your CSL Behring Associate Director of Corporate Accounts.




